Vietnam begins human trials of Covid vaccine Nanocovax
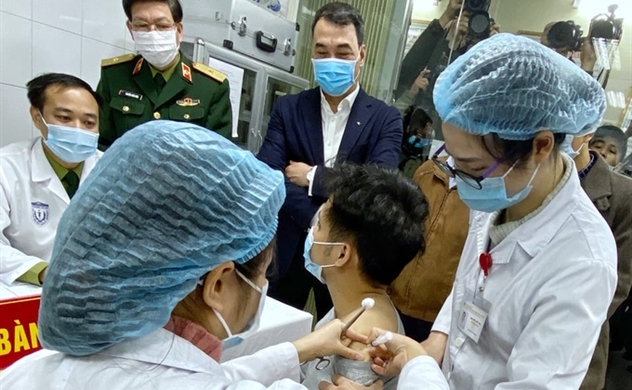
A healthworker gives the first shot in the first-stage safety study of Nanocovax - one of the four made-in-Viet Nam Covid-19 vaccines in Ha Noi on Thursday. Photo: VnExpress
The first three volunteers will stay at the Military Medical Academy for health surveillance within the next 72 hours.
Last week, 60 volunteers were recruited for the first phase of human trails. These volunteers will then be divided into three smaller groups, each receiving three different vaccine dosages: 25 milligrams, 50 milligrams and 75 milligrams.
Each volunteer will receive two shots of the vaccine, with a gap of 28 days between the first and the second in the phase 1 trials.
Currently, Vietnam has four Covid-19 vaccines under development by Nanogen Pharmaceutical Biotechnology JSC, the Institute of Vaccines and Medical Biologicals (IVAC), Vaccine and Biological Production Company No. 1 (Vabiotech) and the Center for Research and Production of Vaccines and Biologicals (Polyvac).
The Nanogen Pharmaceutical Biotechnology JSC is a leading company that is doing research and development of active Biopharmaceutical ingredients (APIs), and specific therapeutic injections in Asia-Pacific region, based on advance in recombinant DNA and protein technologies.
Nanogen’s team consists of high-qualified scientists and graduates in diversity of biotechnology, pharmaceutical and healthcare.
So far, Vietnam has confirmed 1,405 Covid-19 infection cases, including 1,252 recoveries and 35 deaths.
Related to recent outbreak in Ho Chi Minh City, authorities have lifted a ban on several areas traced back to recent community-based COVID-19 cases, as all people having contact with the patients have completed their 14-day quarantine period.
The Ho Chi Minh City Center for Disease Control confirmed on Wednesday morning that no local infections have been detected in the southern metropolis since December 2.
Source: VGP

 TIẾNG VIỆT
TIẾNG VIỆT 




_131447820.png)










